A new low-cost easily scalable medical ventilator is designed and developed by Vega Innovations, a subsidiary of CodeGen Group of Companies. This is now been tested in Sri Lanka under the supervision of Dr Chandana Karunarathna, Consultant in Anesthesiology, Intensive Care and Pain Medicine at Ministry of Health of Sri Lanka
Why invent a low-cost medical ventilator
As the Coronavirus infected patient count rises daily, engineering teams from around the globe are called to action to build emergency medical ventilators to treat the critically ill patients. Vega team answering to this call and recognizing the urgent need to develop local technology to help save lives, put together a small team to develop a medical ventilator in 10 days.
Vega’s low-cost solution enables easy deployment and providing this vital facility of using a ventilator to save a life of a COVID-19 patient with minimum cost and rapid manufacturing as it is engineered 100% in Sri Lanka, with 24/7 hardware and software service teams ready to assist in the fight against Coronavirus.
How is Vega’s Low-cost Medical Ventilator Different from Ambu Bag Design
It is believed that hand-operated bag-valve resuscitator or Ambu bag type is the simplest form of medical ventilators that can be used in a situation such as what we are facing today. However, the team of engineering experts, scientists and medical professionals in the front-line tackling the daily challenges faced in Sri Lanka are testing this much-advanced solution developed bottom up to make it as effective as possible to treat patients at a time where the virus outbreak is reaching its peak.
This particular medical ventilator is designed for treating Coronavirus patients in their critical phase, operates in Volume Controlled – Continuous Mandatory Ventilation (VC-CMV) mode. It takes into the account the drawbacks of the more popular Ambu bag-based design and adds vital features needed to revive Coronavirus patients at full-tilt.
The design is also scalable and can be developed locally in any country by implementing the technology and design architecture to be manufactured under USD 650 per unit.
Specifications of the Low-cost Medical Ventilator
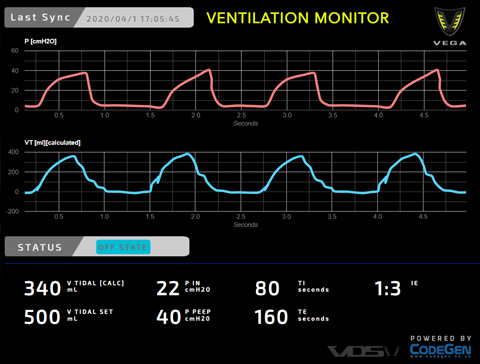
- Volume Controllable From 300ml to 1300ml
- Control Inhalation Timing And 1:E Ratio
- Up To 35 Breath Cycles Per Minute
- PEEP Pressure Adjustable From 30-40 cmH2O
- Inhalation Pressure Up to 40cmh2o
- Safety Valve at 40 cmH2O
- Safety Alarms for Under And Over Pressure As Well Unreachable Breath Cycles
- Runs on Compressed O2 And Compressed Air

Team Behind the Design and Engineering
This project is coordinated by Trace, bringing key stakeholders together to develop, manufacture, and deploy critical medical ventilators to help fight the Coronavirus. The product design and engineering is powered by Vega Innovations. During a short period of time, the engineering teams have developed and now testing the unit with the Health Ministry to take things forward. The first prototype was completed using rapid prototyping tools such as a 3D printer, PCB milling machine, laser cutter and CNC.
Taking it to Production
Once the prototype is thoroughly tested and medical specialists review and approve the design, functionality and deployment of units to local public and private hospitals, the medical ventilator will be manufactured as per industry standard for quick deployment. This is a process done together with the Ministry of Health and backed by CodeGen to mass manufacture the product under the Vega Innovations brand.
CodeGen together with Vega Innovations, are on a mission to transform industries with cutting-edge, high-performance technologies while connecting people, uplifting livelihoods, and advancing the future generations to take ownership of the change they can create with their talent and skills. We will continue to drive change, push boundaries to solve real-world problems globally.
All information are as published on www.vega.lk. Please contact Vega for more information.